[INVNT GROUP]™ unveils strategic acquisitions of 4 historied live event production companies1/1/2022 KUALA LUMPUR, Dec 30 -- [INVNT GROUP] THE GLOBAL BRANDSTORY PROJECT™ has announced the strategic acquisitions of four historied live event production companies: ITP LIVE, InSync Production Services, Thunder Audio, and Morpheus Lights.
Led by President and Chief Executive Officer, Scott Cullather, [INVNT GROUP] represents a growing portfolio of complementary disciplines designed to help forward thinking brands everywhere, impact the audiences that matter anywhere, according to a statement. “The dedicated search for these four companies has been in development for over 18 months. These acquisitions will expand our innovation capabilities, allowing INVNT, our live events business, to offer our customers the ultimate in live events and experiential activations,” said Cullather. [INVNT GROUP] consists of modern brand strategy firm, Folk Hero; creative-led culture consultancy, Meaning; branded content studio and content marketing agency HEVĒ; collegiate events and experiences, INVNT Higher Education, the original global live brand storytelling agency, INVNT and now ITP LIVE consisting of: InSync Production Services, Thunder Audio and Morpheus Lights. The companies will continue to operate under their current names and service their existing roster of clients, seek new clients, and provide services to synergistic INVNT opportunities and projects. Rooted in experiential marketing, [INVNT GROUP]’s expansion comes as part of a strategic, yet natural evolution. This business partnership creates a unique offering in the market, and enhances the group’s abilities to fulfill creative, technological, and strategic designs in dynamic new ways. This transaction was structured by Wolf Karbe, CFO of [INVNT GROUP] and represented by Evergreen Advisors Capital and Center Rock Advisors. Financial terms of the transaction were not disclosed. More details at www.invntgroup.com. -- BERNAMA
0 Comments
BALLERUP, Denmark, Dec 29 (Bernama-BUSINESS WIRE) -- LEO Pharma A/S, a global leader in medical dermatology, announced today that the U.S. Food and Drug Administration (FDA) has approved Adbry™ (tralokinumab) for the treatment of moderate-to-severe atopic dermatitis in adults 18 years or older whose disease is not adequately controlled with topical prescription therapies or when those therapies are not advisable. Adbry can be used with or without topical corticosteroids.¹ Adbry is the first and only FDA approved biologic that specifically binds to and inhibits the IL-13 cytokine, a key driver of atopic dermatitis signs and symptoms.¹,²,³ “Today’s FDA approval of Adbry is a major milestone for LEO Pharma and for the millions of people living with moderate-to-severe atopic dermatitis who struggle to find a suitable treatment option for this chronic and debilitating disease,” said Anders Kronborg, Chief Financial Officer and Acting Chief Executive Officer of LEO Pharma A/S. “As our first biologic in the U.S., Adbry signifies important progress in our mission of advancing the standard of care in medical dermatology.” The approval of Adbry is based on safety and efficacy results from the ECZTRA 1, 2 and ECZTRA 3 pivotal Phase 3 trials, which included nearly 2,000 adult patients with moderate-to-severe atopic dermatitis.¹ Safety data was evaluated from a pool of five randomized, double-blind, placebo-controlled trials, including ECZTRA 1, 2 and ECZTRA 3, a dose-finding trial, and a vaccine response trial.¹ “Atopic dermatitis can be severe and unpredictable, which makes it not only challenging for patients to achieve long-term disease control, but also for clinicians to treat, since there are limited treatment options for this burdensome chronic skin disease,” said Jonathan Silverberg, MD, PhD, MPH, Associate Professor of Dermatology at George Washington University School of Medicine and Health Sciences, and tralokinumab clinical trial investigator. “Adbry will be an important addition to our therapeutic armamentarium as a treatment designed to specifically target and neutralize the IL-13 cytokine, thereby, helping patients manage their atopic dermatitis.” Adbry will be available in a 150 mg/mL prefilled syringe for subcutaneous injection with an initial dose of 600 mg followed by 300 mg every other week. Adbry can be used with or without TCS.¹ A dosage of 300 mg every four weeks may be considered for patients below 100 kg who achieve clear or almost clear skin after 16 weeks of treatment.¹ To help eligible patients have access to Adbry, LEO Pharma will introduce the AdbryTM AdvocateTM Program to support U.S. patients at diagnosis and through treatment with Adbry. “For people living with atopic dermatitis, the experience goes beyond the skin, often impacting important psychosocial aspects of their life,” said Julie Block, President and CEO of the National Eczema Association. “It’s exciting to see a new, targeted therapeutic option for adult patients living with moderate-to-severe atopic dermatitis. Therapeutic advances like this provide much needed hope for those who may have spent years struggling to find a suitable therapy to alleviate the burden of this disease.” The FDA approval marks the fifth global regulatory approval for tralokinumab in 2021. Tralokinumab is marketed outside of the U.S. under the tradename Adtralza® and is currently approved in the European Union, Great Britain, Canada and the United Arab Emirates. http://mrem.bernama.com/viewsm.php?idm=42051  The scene for releasing achievements on construction of international tropical farm produce indices publishing center in Hainan. BEIJING, Dec. 28, 2021 /Xinhua-AsiaNet/-- Three annual reports on natural rubber price indices, banana industry development and agarwood industry climate index were released on the 2021 Boao Forum for Entrepreneurs held in Hainan, a coastal island province in South China recently. The three reports, namely Xinhua-Hainan State Farms (HSF) China natural rubber series price index report (2020-2021), China banana industry development report (2020-2021) and China agarwood industry climate index report (2021), are typical achievements of the province in crafting itself into an international tropical farm produce indices publishing center. Since 2018, the province has been required to construct an international transaction, pricing and price indices releasing center for tropical farm produces including mainly the natural rubber. From then, Xinhua-run China Economic Information Service, the People's Government of Chengmai County in Hainan Province and Hainan State Farms Investment Holdings Group Co., Ltd. have jointly incubated a series of price indices for a dozen of tropical farm products such as natural rubber, banana, pepper, and agarwood. With the indices family, tropical farm produce digitalization and brand construction forged ahead in Hainan and helped the province build its influences and advantages in international tropical agriculture and fostered high-quality development of the Hainan Free Trade Port. As the Xinhua-Hainan State Farms (HSF) China natural rubber series price index report (2020-2021) tells, natural rubber prices generally fluctuated at high levels during 2020 and 2021 due to the climbing shipping costs and operation conditions of downstream factories amid the global COVID-19 pandemic, production area climate change and bulk commodity market volatility. In the coming production season in 2022, industry players shall pay extra attention to the influences of La Nina phenomenon on progresses of cutting in natural rubber production areas to judge the price trends. According to the China banana industry development report (2020-2021), China's banana imports underwent structural changes, with imports in border areas of the country down 28.2 percent year on year, sales recovery of bananas in northern Myanmar unblocked and imports of bananas from Cambodia up notably. Under such circumstances, the report suggests that banana industry in Chinese provinces, in particular Hainan to actively seize opportunities to beat drum for homegrown bananas by enhancing industry chain standards construction and quality control. The China agarwood industry climate index report (2021) showed that consecutive breakthroughs in plantation and producing techniques have shorten the cycle of agarwood production in recent years. When the agarwood industry size keeps expanding, the industry needs integrated development and establishment of product supply chain tracing system to promote products standardization and boost brand effects. Source: China Economic Information Service Image Attachments Links: Link: http://asianetnews.net/view-attachment?attach-id=411869 Caption: The scene for releasing achievements on construction of international tropical farm produce indices publishing center in Hainan. - Successful nomination of lead SAFEbody candidates triggers milestone payment -
SAN DIEGO and SUZHOU, China, Dec 23 (Bernama-GLOBE NEWSWIRE) -- Adagene Inc. (“Adagene”) (Nasdaq: ADAG), a biopharmaceutical company committed to transforming the discovery and development of novel antibody-based immunotherapies, today announced achievement of a key milestone in its ongoing collaboration with Exelixis for development of novel masked antibody-drug conjugate (ADC) candidates leveraging Adagene’s proprietary SAFEbody precision masking technology. Under the terms of a collaboration and licensing agreement established in early 2021, Adagene will receive a $3 million milestone payment for successful nomination of lead SAFEbody candidates for one of its collaboration programs. “We are extremely proud of successfully providing lead SAFEbody candidates to Exelixis following the start of our collaboration earlier this year. Exelixis’ selection of these lead candidates further validates our proprietary SAFEbody technology and highlights the prowess of our overall DPL platform,” said Peter Luo, Ph.D., Co-founder, Chief Executive Officer, and Chairman of Adagene. “Our SAFEbody masking technology is receiving clinical validation in our ongoing clinical trial from our anti-CTLA-4 monoclonal antibody program, and also marks a potential paradigm shift in the way a highly potent modality such as ADCs can be designed and developed, ultimately helping to improve the lives of patients suffering from cancer and other debilitating diseases.” Under the terms of the agreement, Adagene received an upfront payment of $11 million and Exelixis can nominate two targets for development of SAFEbody candidates during the collaboration. Adagene is eligible for development and commercialization milestones, as well as royalties on net sales of products developed around each of these targets. SAFEbody technology is designed to overcome safety and tolerability challenges associated with many antibody therapeutics by using precision masking technology to shield the binding domain of the biologic therapy. This allows for improved tumor-specific targeting of antibodies, while minimizing on-target off-tumor toxicity in healthy tissues, a longstanding challenge with many antibody therapeutics. In addition to ongoing collaborations, Adagene also applies its SAFEbody technology to develop candidates for its wholly-owned deep, broad and differentiated pipeline. These include ADG126, an anti-CTLA-4 SAFEbody in phase 1 dose escalation as monotherapy, as well as five highly differentiated programs in IND-enabling studies such as ADG153, an anti-CD47 SAFEbody using the potent IgG1 isotype, and ADG152, anti-CD20xCD3 bispecific POWERbody™ T-cell engager. A total of five antibodies are in clinical development by Adagene and its partners, leveraging the company’s AI-driven antibody discovery and development platform. About Adagene Adagene Inc. (Nasdaq: ADAG) is a platform-driven, clinical-stage biopharmaceutical company committed to transforming the discovery and development of novel antibody-based cancer immunotherapies. Adagene combines computational biology and artificial intelligence to design novel antibodies that address unmet patient needs. Powered by its proprietary Dynamic Precision Library (DPL) platform, composed of NEObody™, SAFEbody®, and POWERbody™ technologies, Adagene’s highly differentiated pipeline features novel immunotherapy programs. Adagene has forged strategic collaborations with reputable global partners that leverage its technology in multiple approaches at the vanguard of science. For more information, please visit: https://investor.adagene.com. SAFEbody® is a registered trademark in the United States, China, Australia, Japan, Singapore, and the European Union. http://mrem.bernama.com/viewsm.php?idm=42034 HVDC Light® will connect low-carbon power from the mainland grid to ADNOC’s production operations as a strategic project to enable a sustainable, flexible and secure power supply.
Zurich, Switzerland, Dec 23 (Bernama-GLOBE NEWSWIRE) -- Hitachi Energy today announced it has won a major order from Samsung C&T Corporation, one of the world’s largest engineering and construction companies, to connect ADNOC’s offshore operations to the onshore power grid in the United Arab Emirates owned and operated by Abu Dhabi National Energy Company PJSC (TAQA). Hitachi Energy’s HVDC Light® technology and MACHTM digital control platform1 will enable the transfer of cleaner and more efficient power from the mainland to power ADNOC’s offshore production operations, enabling a carbon footprint reduction of ADNOC’s offshore operations by more than thirty percent. This innovative solution reinforces Hitachi Energy’s commitment to helping customers and countries to transition towards a carbon-neutral future and help enable the ‘2050 Net-Zero Initiative’ of the UAE. With a capacity of 3,200 megawatts (MW), the two HVDC links will be by far the most powerful power-from-shore solution in the Middle East and North America (MENA) region to date. It is also the first HVDC power-from-shore solution outside Norwegian waters. This innovative solution reflects how Hitachi Energy continues to pioneer technology to address the growing interest from national and independent oil and gas companies to power their offshore production facilities with carbon-free energy from onshore power grids. “We are proud to be enabling Abu Dhabi and ADNOC to make significant progress on their pathway toward achieving the United Arab Emirates’ ambition to be carbon-neutral by 2050,” said Claudio Facchin, CEO of Hitachi Energy. He continued, “At Hitachi Energy we are championing the urgency of the clean energy transition, and this major order is further evidence that we are a ‘go to’ partner for developing and deploying technologies and solutions that are advancing the world’s energy system to be more sustainable, flexible and secure.” Mr. SH Kim, Procurement Manager at Samsung C&T Corporation, commented, “In Hitachi Energy, we have selected a trusted partner who brings deep global competence and a strong mindset of collaboration and innovation.” SH Kim continued, “Together, we will serve ADNOC with pioneering technologies that are proven to deliver for such a large HVDC project.” The entire power-from-shore project will comprise two HVDC power links, which will connect two clusters of offshore oil and gas production facilities to the mainland power grid, a distance of up to 140 kilometers for each cluster. Hitachi Energy is supplying four converter stations, which convert AC power to DC for transmission in the subsea cables, then reconvert it to AC from DC for use in the offshore power systems. The HVDC technology will be supplied from Hitachi Energy’s global competence centers. Also included in the order are system studies, design and engineering, supply, installation supervision and commissioning. Hitachi Energy will support the customers with a long-term life-cycle service agreement leveraging digital technologies to ensure system availability and reliability over the HVDC links’ long operating life. HVDC Light is a voltage source converter technology that was pioneered by Hitachi Energy. It is the preferred technology for many grid applications, including interconnecting national power grids, integrating offshore wind parks with mainland transmission systems, feeding more power into congested city centers, interconnecting asynchronous networks that operate at different frequencies, and power from shore. HVDC Light’s defining features include uniquely compact converter stations (which is extremely important in space-critical applications like offshore wind, offshore production facilities and city-center infeeds), exceptionally low electrical losses, and black-start capability to restore power after a grid outage. Hitachi Energy pioneered commercial HVDC technology almost 70 years ago and has delivered more than half of the world’s HVDC Classic projects and more than 70 percent of the world’s voltage source conversion HVDC projects. Notes: 1. Modular Advanced Control for HVDC (MACH™) 2. The estimated reduction in carbon footprint is based on Hitachi Energy’s own calculations. About Hitachi Energy Hitachi Energy is a global technology leader that is advancing a sustainable energy future for all. We serve customers in the utility, industry and infrastructure sectors with innovative solutions and services across the value chain. Together with customers and partners, we pioneer technologies and enable the digital transformation required to accelerate the energy transition towards a carbon-neutral future. We are advancing the world’s energy system to become more sustainable, flexible and secure whilst balancing social, environmental and economic value. Hitachi Energy has a proven track record and unparalleled installed base in more than 140 countries. Headquartered in Switzerland, we employ around 38,000 people in 90 countries and generate business volumes of approximately $10 billion USD. # # # Attachment · Hitachi Energy HVDC Light Valve Hall Rebecca Bleasdale Hitachi Energy Ltd. +41 78643 2613 [email protected] Source: Hitachi Energy Ltd. --BERNAMA Rancho Santa Margarita, Calif., Dec 22 (Bernama-GLOBE NEWSWIRE) -- Blue California, the producer of natural science-based ingredients, provides FineCap™ a comprehensive microencapsulation technology platform, equipped with 20 microencapsulation technologies, to deliver active ingredients and satisfy our clients’ needs.
Microencapsulation is the process in which tiny particles of solid, liquid, or gas are packaged within a matrix to form a capsule. The capsule is coated with a protective layer to avoid degradation from exposure to environmental factors such as water, oxygen, heat, and light. “Brands that seek to expand their products’ qualities and boost their product portfolios will find many benefits to the FineCap platform,” said Dr. Cuie Yan, vice president of encapsulation. “FineCap takes microencapsulation a step further by offering a variety of technologies and targeting customers’ specific needs in tackling active ingredients with unique characteristics, such as strong odor, taste or stability problems that challenge formulators.” Microencapsulation systems have been widely used across multiple industries, including the pharmaceutical, food, supplement, personal care, and fragrance industries, for active ingredients like medicines, flavors/fragrances, polyunsaturated fatty acids, probiotics, natural pigments, vitamins, antioxidants, etc. Space agency NASA also uses encapsulation technologies for spacecraft. The pharmaceutical industry uses microencapsulation often to control the release of active pharma ingredients (API). Blue California has created the FineCap platform to serve customers’ growing demands for better performance of API, functional ingredients, dietary supplements, flavors, fragrances, cosmetics, and personal care products. For example, FineCap protects API from degradation, unpleasant tastes or aroma, and maintains its efficacy, by controlling its release. FineCap enables flavors to thrive in food and beverages with integrity, intensity, and extended shelf-life. In fragrances, FineCap guarantees brands to control the precise fragrance release rate, location, and duration. Personal care products benefit from FineCap by protecting the delicate top-notes and cosmetic actives from oxygen, moisture, temperature, and light deterioration. A more comprehensive look into the benefits that FineCap delivers in these product segments can be found here. “Our comprehensive FineCap platform has been serving and supporting formulators looking to launch market-winning products with better qualities and shelf-life that consumers are seeking,” said Dr. Yan. “We’re enabling brands to quickly create products from innovative concepts, benchtop development, to pilot and full commercial manufacturing, with improved efficacy, taste, color, texture, and shelf life, along with vegan, organic, or Halal certificate.” The FineCap platform investment builds on Blue California’s 25-year legacy of producing botanical extracts and now natural flavors and fragrances and focuses on developing sustainable ingredients made through bioconversion or fermentation. ### About Blue California Blue California is an entrepreneurial, science-based solutions provider and manufacturer of clean, natural, and sustainable ingredients used in food, beverage, flavor, fragrance, dietary supplements, personal care, and cosmetic products. For more than 25 years, Blue California has built a strong reputation for creating value in these diverse natural products and nature-inspired industries. Attachment Ana Arakelian Blue California +1-949-635-1991 [email protected] SOURCE: Blue California Ingredients http://mrem.bernama.com/viewsm.php?idm=42019 KUALA LUMPUR, Dec 21 (Bernama) -- In the aftermath of the most devastating flood disaster in Malaysian history, Nitesh Malani, Chairman of Yayasan Usahawan Malaysia and founder of the SME and Entrepreneurs Business Awards (SEBA), urges the government to provide a moratorium on bank loan repayment for SMEs to ease the burden of these businesses in their recovery period.
After a lengthy period of business shutdown and financial constraints due to the COVID-19 pandemic, businesses are beginning to recover, only to be struck by another tragedy. The flood has impacted an estimated of up to 18,000 SMEs and entrepreneurs, with the majority reporting significant damage to cars, machinery, and inventories, many of which cost millions of ringgit. Bank Negara Malaysia (BNM) has established the Disaster Relief Facility 2022 with a total of RM 200 million fund allocation to help alleviate the financial burden of MSMEs affected by the flood and assist them in resuming business operations. BNM grants up to RM 500,000 per SME and RM 75,000 per micro-enterprise. Effective December 27, 2021, businesses and entrepreneurs may start applying for this assistance until further notice. The financing period of up to five years comprises a six-month moratorium on principal and interest/profit payments. Other government initiatives to aid flood-affected SMEs include RM5,000 in interest-free funding from Bank Simpanan Nasional and RM30 million in loans from Tekun Nasional. "I urge the Ministry of Entrepreneur Development and Cooperatives (MEDAC) to develop an immediate strategy to aid and assist these SMEs in their recovery from what appears to be the country's most devastating flood calamity. In addition to the funds provided by Bank Negara Malaysia, I urge the government to go a step further and propose an interest-free loan repayment moratorium for affected SMEs. The government must develop a strategic planning team to address and alleviate the suffering of SMEs during these difficult times. Only with this precise strategic team in place will they be able to plan the next strategy for aiding these SMEs and entrepreneurs. More needs to be done to assist MSME and prompt action my key agencies and private financial institutions needs to be executed. It's about livelihood of thousands as most people have lost everything including savings. Bureaucracy needs to be removed in this moment of need more importantly to support all effected businesses. Yayasan Usahawan Malaysia is willing to assist businesses in whichever manner we can," Nitesh says. YUM will commence an immediate effort in reaching out to as many businesses that requires assistance via a specific hotline and MSME can also connect with us via [email protected] http://mrem.bernama.com/viewsm.php?idm=42018 PRESTIGE BIOPHARMA COMMENCES CONSTRUCTION OF INNOVATIVE DISCOVERY CENTER (IDC) IN BUSAN, KOREA21/12/2021  SINGAPORE, Dec 20 (Bernama-BUSINESS WIRE) -- Prestige BioPharma Limited (PBP, 950210: KRX), a Singapore-based biopharmaceutical with operations in USA and South Korea, commences construction of Innovative Discovery Center (IDC) in Busan, South Korea. The construction of IDC is scheduled for completion by February 2023. This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20211219005014/en/ PBP holds a groundbreaking ceremony on December 21, 2021 with Korean government officials and guests including Deputy Mayor of Busan City Mr. Yoon-il Kim, Director General for Investment Policy of the Ministry of Trade, Industry and Energy Mr. Jong-young Jung and Head of Development Administration Division in Busan-Jinhae Free Economic Zone Authority (BJFEZ) Mr. Seokho Cha. PBP is on target to build the global-scale R&D centre with a total area of approximately 34,000 square meters and facilities sufficient for 800 researchers. Earlier in May this year, PBP and Busan City Government had signed an MOU for the establishment of IDC in which the company’s plan to invest over US$200 million by 2030 was announced. IDC will be focusing on diverse first-in-class antibody biologics discovery starting from 8 bispecific antibody projects based on PBP’s proprietary oncology therapeutic targets, PAUF and CTHRC1. Next generation vaccines using mRNA technologies and bioinformatics will be developed in IDC for current and potential future pandemics. IDC also will be hiring more than 250 of PhD and highly qualified R&D personnel over the next five years and fifty percent of its R&D resources will be sourced from local universities and community. As the first global scale R&D center in Busan, IDC is expected to lead the growth of Busan’s bioindustry and to lay a foundation to make the region into a viable bio-cluster that supports the local economy and drives mutual growth in academia and industry. IDC is planning to implement an open innovation platform that provides the local biomedical society a ‘one stop’ R&D solution for full cycle of new drug development targeting global markets. PBP’s CEO, Dr Lisa S. Park commented: “IDC will be leading PBP Group’s innovative drug discovery R&D in connection with our research centers in Singapore and US and creating a global open innovation hub for industry-academic collaboration” and “we envision IDC where Prestige Biopharma’s innovation realizes and the start of a new world-class bio-cluster here in Busan”. Hyeong-joon Park, Mayor of Busan City, commented: “We are very excited to have a global scale bio-R&D center in Busan and work with a global leading bio-pharmaceutical company, Prestige Biopharma. Busan City will fully support PBP’s IDC so this novel partnership can lead a local bio-industry’s growth and contribute to local economic development and high-quality job creation”. http://mrem.bernama.com/viewsm.php?idm=41993 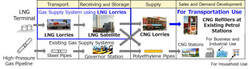 KUALA LUMPUR, Dec 20 (Bernama) -- Osaka Gas Co Ltd (Osaka Gas) has announced participation in the City Gas Distribution (CGD) business in India through its wholly-owned subsidiary Osaka Gas Singapore Pte Ltd (OGS) by investing in AG&P CGD HoldCo SPV3 (Singapore) Pte Ltd (AG&P SPV3), together with Japan Overseas Infrastructure Investment Corporation for Transport & Urban Development (JOIN). AG&P City Gas is developing the 12 CGD geographical areas it was awarded in South India and Rajasthan. Osaka Gas is the first Japanese company to participate in the CGD sector in India through this investment. Based on a statement, as a successful bidder for the city gas development projects supported by the Government of India, AG&P City Gas obtained exclusive gas sales rights and infrastructure ownership rights (exclusive rights) in India for a fixed period of time for its 12 concessions. India promotes greater use of natural gas through the development of city gas infrastructure that supports the wider use of compressed natural gas vehicles (CNG vehicles) in response to the increasing energy demand and a need to reduce carbon emissions and air pollution resulting from economic growth in the country. This city gas venture has been developed by the AG&P Group led by AGP International Holdings Pte Ltd (AGPIH), AG&P SPV3’s parent company, in which Osaka Gas also invests in. With exclusive rights for a vast swath of southern India, roughly equivalent to ¾ the area of Japan, the venture aims to develop into a business in a scale of 3.5 billion m³ in gas sales volume, which is approximately half of Osaka Gas’ city gas sales volume in Japan. The venture also plans to promote city gas demand for CNG vehicles as well as for household, commercial, and industrial uses, and utilise LNG lorries as one of the main gas supply system, which is introduced for the first time to India, to reduce the time for the development and roll-out of the city gas infrastructure. AG&P CGD, the operator of this city gas business, is jointly owned by AGPIH, the leading downstream LNG platform and infrastructure development company. Osaka Gas has been expanding its business of gas distribution, gas sales to industrial customers, energy services, rooftop solar power generation in Southeast Asia. -- BERNAMA KUALA LUMPUR, Dec 21 -- Gupshup, the leader in conversational messaging, has announced the launch of a WhatsApp-based commerce solution enabling any business to create a digital ‘storefront’ on WhatsApp.
Businesses can enable conversational journeys across every stage of the buying process: pre-purchase, purchase, and post-purchase. Based on WhatsApp’s recently launched commerce capability, Gupshup can help a business manage the complete buying experience on WhatsApp. With more than two billion active monthly users in 180 countries using WhatsApp, it is the ideal conversational commerce platform for businesses. This full-featured commerce solution also leverages Gupshup's recently launched 1-Click Bill Pay feature, which enables businesses to collect payments via WhatsApp and other messaging apps. With Gupshup’s WhatsApp-based commerce solution, a business can create a product catalogue on WhatsApp, converse with customers via AI-powered chatbots for product discovery, guide them through checkout and payment, and support them 24/7 with chatbots and live agents. Customers can also add preferred items to their carts on WhatsApp, interact with the businesses in real time, and make payment through Gupshup’s recently launched 1-Click Bill Pay link, all without leaving the chat window. “As one of the earliest Solution Provider partners of WhatsApp, we have already helped thousands of businesses across the world go live on WhatsApp Business. “Our knowledgeable sales and solutions teams, along with a network of global partners, help businesses accelerate their digital transformation,” explained Gupshup COO, Ravi Sundararajan in a statement. “We are seeing strong demand for the new Commerce features, and the early results for businesses are promising. This is certainly going to revolutionise the way businesses conduct e-commerce.” Gupshup is present in India, LATAM, South East Asia, Middle East, Eastern Europe, Africa and the United States. For more information, visit www.gupshup.io. -- BERNAMA |
Archives
April 2023
Categories |
 RSS Feed
RSS Feed
